Dr. Martin Presents at THT 2026 in Boston: From Hippo Pathway Discovery to First-in-Human Gene Therapy
Dr. returned from Boston this week, where he presented at THT 2026: Technology and Heart Failure Therapeutics, held March 2–4 at the Omni Boston Hotel at the Seaport. Organized by the Cardiovascular Research Foundation, THT has become one of the premier international forums for advancing cutting-edge treatments in heart failure, and this year's program features 24 late-breaking clinical science presentations, first-in-human studies, and sessions spanning AI, advanced LVAD technologies, and cardiac xenotransplantation. Dr. Martin's talk, "YAP Induces Cardiomyocyte Proliferation," traces the arc of our lab's research from fundamental discovery through clinical translation, a journey that has culminated in the first-ever clinical trial of a gene therapy designed to regenerate human heart tissue.
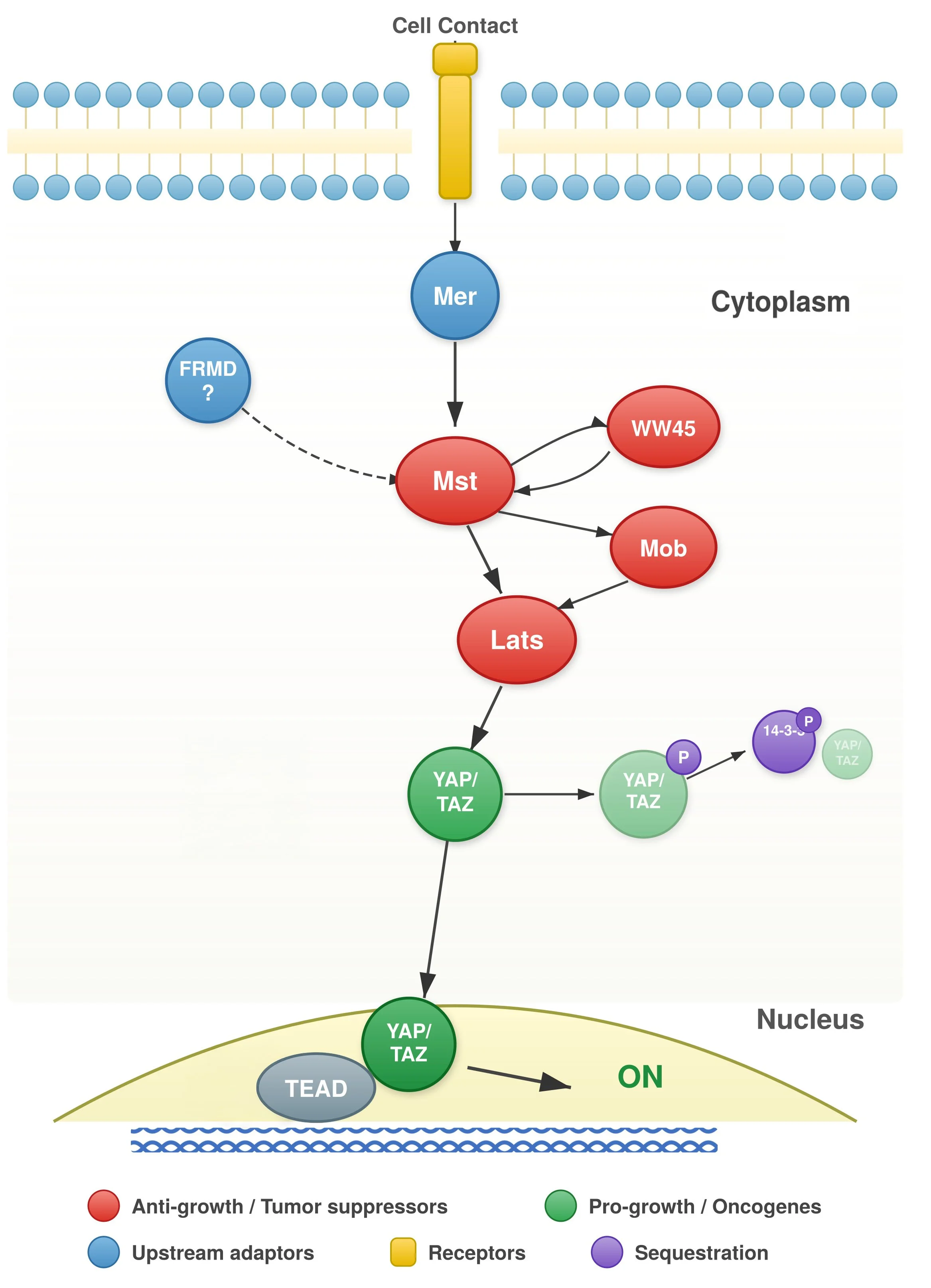
Hippo: Unlocking the Heart's Own Repair Mechanism
At the center of our work is the Hippo signaling pathway, a highly conserved kinase cascade that phosphorylates and inhibits the transcriptional co-activator YAP. In the adult heart, active Hippo signaling essentially acts as a brake on cardiomyocyte proliferation, which is why the human heart replaces barely 1% of its muscle cells per year and cannot meaningfully repair itself after injury. We made the landmark discovery that deleting components of the Hippo pathway in adult cardiomyocytes can reverse this limitation. In mouse models of established ischemic heart failure, deletion of the Hippo pathway induces reverse remodeling: scarred, failing hearts showed restored architecture and significantly improved cardiac function. These findings demonstrated something remarkable: a failing heart could be coaxed into regeneration. Subsequent work revealed what actually happens inside these regenerating hearts at the cellular level. When YAP is activated, dormant heart muscle cells wake up and divide to create new cells, something the adult heart almost never does on its own. Using advanced genomic mapping tools, we identified a special regenerative state in which heart muscle cells temporarily disassemble their internal machinery and shift how they produce energy, essentially reverting to a more youthful form before multiplying. Perhaps most importantly, these regenerating heart cells don't work alone. They recruit nearby support cells, including specialized fibroblasts and immune cells, to create what the team calls a "pro-renewal triad," a local environment that appears to be essential for successful heart muscle regeneration.
From Bench to Bedside: The SALVADOR-HF Trial
The translational potential of these discoveries is now being tested in humans. Dr. Martin co-founded Medley Therapeutics (formerly YAP Therapeutics) to develop YAP101, a first-in-class AAV9-based gene therapy that uses a cardiomyocyte-specific promoter to express short hairpin RNA (shRNA) targeting Salvador 1 (SAV1), a key component of the Hippo pathway. By transiently suppressing Hippo signaling, YAP101 is designed to activate YAP and trigger self-limiting myocardial regeneration. Preclinical studies in a pig model of myocardial infarction demonstrated that SAV knockdown improved heart function and reduced fibrosis, results published in Science Translational Medicine in 2021. These findings supported the initiation of the Phase 1 SALVADOR-HF clinical trial (NCT06831825), an open-label, dose-escalation study evaluating the safety and preliminary efficacy of YAP101 in adults with ischemic heart failure and reduced ejection fraction. The trial is being conducted at the Texas Heart Institute at Baylor College of Medicine, with the therapy delivered via transendocardial injection during cardiac catheterization. In November 2025, Medley Therapeutics announced that the first cohort of three patients had been successfully dosed, and the independent Safety Review Team recommended escalation to the next dose level, an important early milestone for the program.
The Bigger Picture: Cardiomyocyte Regeneration as a Series of Discrete Events
A key theme of Dr. Martin's presentation is that cardiac regeneration is not a single switch but a coordinated sequence of discrete events: dedifferentiation and sarcomere breakdown, proliferation to generate new cardiomyocytes, an inflammatory response involving macrophages, metabolic reprogramming, and ultimately redifferentiation into functional heart muscle. Understanding each step and how they interact is essential for optimizing regenerative therapies. This framework positions the Martin Lab's work at the intersection of basic biology and clinical innovation, exactly the kind of translational science that THT was designed to spotlight.
For more information about our lab’s research, visit jfmartinlab.com/research. For details on the SALVADOR-HF trial, see ClinicalTrials.gov (NCT06831825). Learn more about THT 2026 at thtmeeting.com.